HELIUM | emission | one of each color except cyan/green
HYDROGEN | emission | one of red, cyan, indigo, violet.
INCANDESCENT | continuous | all visible
FLUORESCENT | continuous | all visible
MYSTERY LAMP | emission | one of red, cyan, indigo, violet
SOLAR | continuous | all visible
1. The mystery gas was hydrogen.
2. Mercury vapor.
3. Red filter: We saw all the colors still, but you're only supposed to see red (it blocks out all other colors except red) | Blue filter: Same. We saw all, only supposed to be blue. | Green: We couldn't find a green filter, but one would assume the results would be similar.
4. Neon lights of different colors do not contain neon: true neon glows red, but often neon lights are made with other gases that either glow different colors or tint the glass a different color with a white glowing gas.
5. Every wavelength has some absorption lines, but they're small pieces- one must consider that the light is travelling through millions of miles as well as the atmosphere.
6.
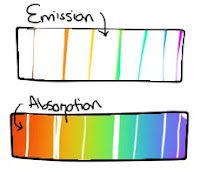
Absorption lines are the tiny lines that are missing when a continuous light source is shone through the cold gas. Emission lines are the lines corresponding to the missing places that are shown when the gas itself is heated and emits light.
7.
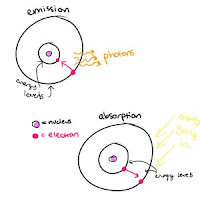
Emission is when the electrons fall to a lower energy level- they emit photons, causing the glowing-ness we see.
Absorption is when the electrons are promoted to a higher energy level, usually caused by an outside source of energy, like heat.
8. Emission lines are discreet and specific wavelengths of light are emitted- in continuum radiation, the light source emits light at all wavelengths.
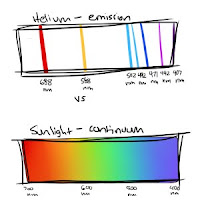
Helium - only certain wavelengths show up.
Sunlight (many, many gases, etc) - all wavelengths show up.
I made all of these little examples myself in Sai, a painting program I use. : ) I didn't just find them on the internet.
This is well done. As part of your reflection: How does this relate to what we are learning? What does it change about what you knew about light?
ReplyDelete